Memorial to Prof. Alfred G. Redfield
Prof. Mary Roberts

Al’s last project was to make an add-on shuttling system for a Varian INOVA Plus (it was underutilized at Brandeis once the 600 MHz spectrometer was available) that was simple enough for a biochemist to use (that was where I came in). The initial goal was ‘high-resolution’ 31P field cycling relaxometry. While there are lots of interesting phosphorus-containing biomolecules, at the high fields of modern spectrometers the 31P T1 has a large CSA relaxation component as well as dipolar interactions. Initially, Al thought that at the very least one would be able to get correlation times and an effective distance between the 31P and 1H nuclei that relax it. I used to refer to it as ‘Al’s Spin Spa’ because shuttling to low fields was a great way to relax spins.
From the very first study on a DNA octamer, he noted that the 31P R1 profiles had interesting features. It was easy to see a dipolar dispersion (the average correlation time of all the 31P , ~ 3-4 ns, corresponded to overall octamer rotation) and a high-field CSA component, but there was also ‘excess’ R1 between the two that was unexpected. Al initially treated it as a CSA relaxation contribution connected to molecular tumbling but admitted it was an ad hoc assumption.
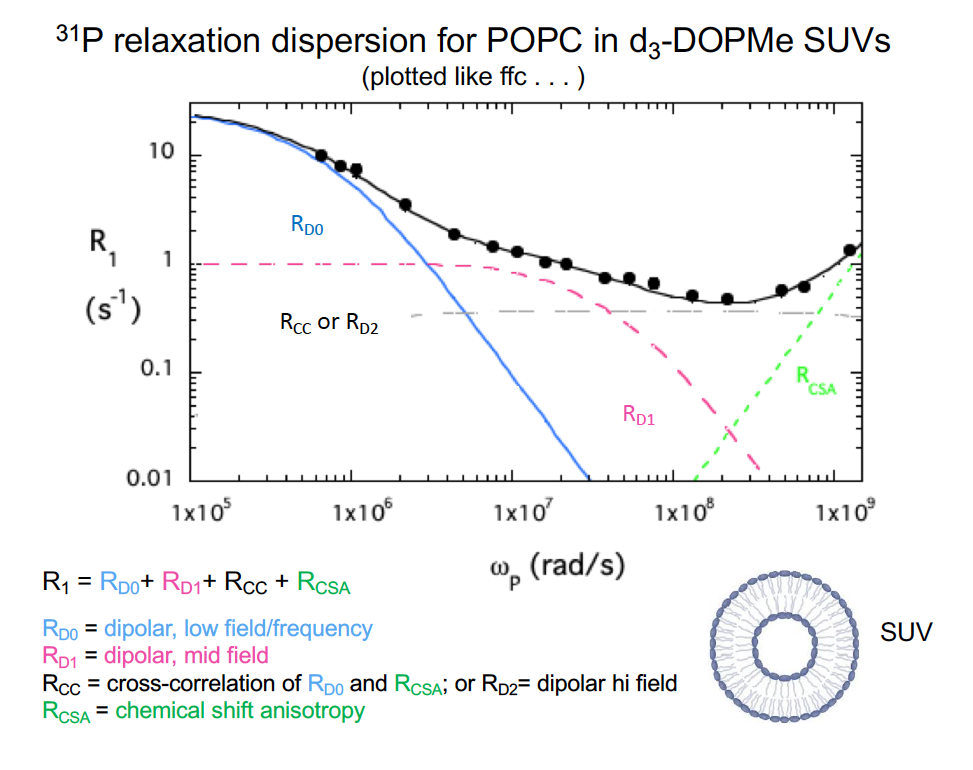
Since I deal with phospholipids in vesicles and micelles, I persuaded Al that we should look at those – preferably as mixtures with multiple types of phospholipids. With the initial pneumatic shuttling system, we could only go down to 0.06 T, and so the basic pattern for the phospholipids was similar to that for the DNA octamer. Except the dipolar interaction we saw had a correlation time ~ 10 ns, not comparable to what is expected for overall tumbling of small vesicles. Al, in tinker mode, decided that we needed to get to even lower fields to see if we could see a dipolar interaction that corresponded to vesicle tumbling. I came back to use it after a few weeks and there was now a computer-controlled Helmholtz coil above the magnet and samples were now shuttled into the middle of it. With accessible fields down to 0.003T, we could see another dipolar dispersion at very low fields with τ ~0.5 μs, that corresponds to what the fast-field cycling people call RMTD (Rotation Mediated by Translational Diffusion). That correlation along with the average size of these small spherical vesicles provides a way of extracting a lateral diffusion coefficient, Dlat for each type of phospholipid in the vesicle. It turns out to be an interesting tool to assess how ‘mixed’ the phospholipids are and what happens when proteins are added.
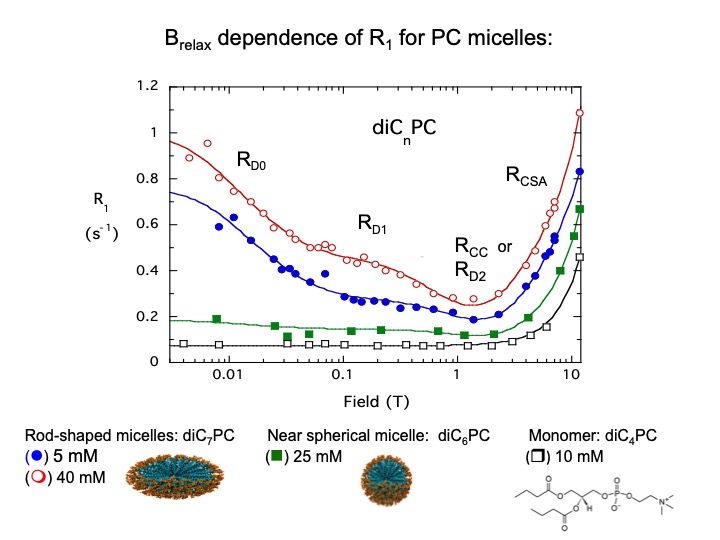
We took advantage of the low-field dipolar relaxation by measuring changes in the magnitude of the relaxation rate extrapolated to zero field, R(0), upon the addition of spin-labeled proteins. The reason to use the very low field region was that an individual phospholipid had to occupy a discrete binding site on the protein fori, at least, a μs for us to see this interaction. By subtracting the relaxation measured for the phospholipids with unlabeled protein, and fitting the remaining ΔR1 we could obtain a correlation time, τP-e, and ΔRP-e(0), the increase specifically caused by proximity to the spin label and we could estimate rP-e. Repeating the experiment with the spin-label on different residues provided enough distance constraints that we could suggest a probable location of a specific lipid binding site. Al’s Spin Spa and his PR1E (paramagnetic R1 enhancement) experiment turned out to be a great way to identify discrete phospholipid binding sites on a protein [Ref: Wei, Stec, Redfield, Weerapana, and Roberts (2015) J. Biol. Chem. 290:1592-606]. Even better, at those low fields we typically used ratios of total phospholipid to protein > 100 (and often much higher). We were not decorating the phospholipid interface with proteins. We used it effectively with micelles as well as vesicles, where one useful property of the method is that it is easy to see if an added protein changes micelle size by measuring the τP-e. With two of the signaling proteins we examined, micellar phosphoinositides were aggregated in a way that suggested particular conformational changes when they were bound to the phospholipids interface.
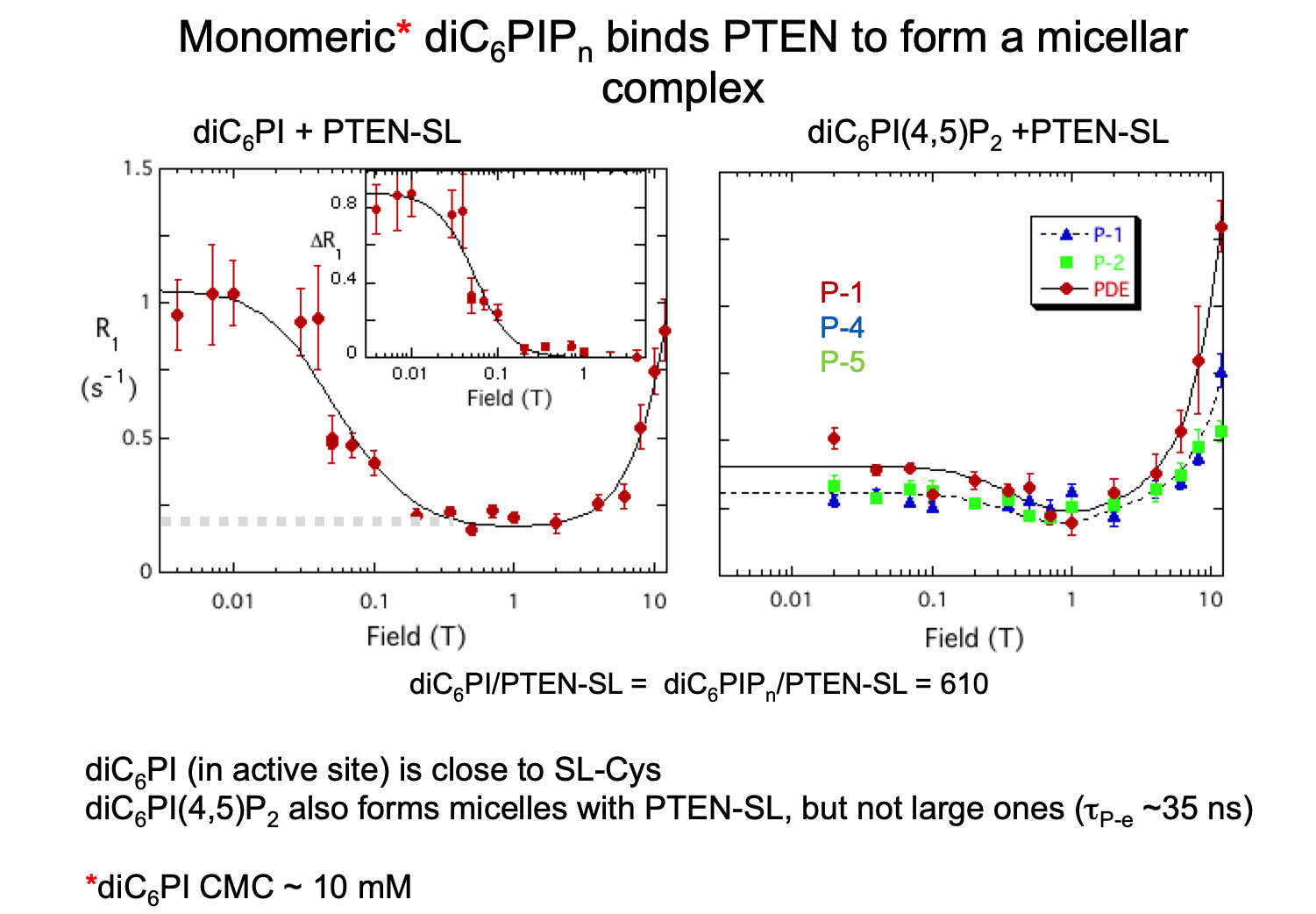
Another advantage of the PR1E experiment is that you can measure weak binding (20-80 mM Kd) for small and medium size molecules. Fast exchange between free and bound is reflected in R1 at a given field, but the dipolar dispersion observed will reflect the correlation time for the complex; its magnitude will be proportional to the fraction of ligand that is bound.
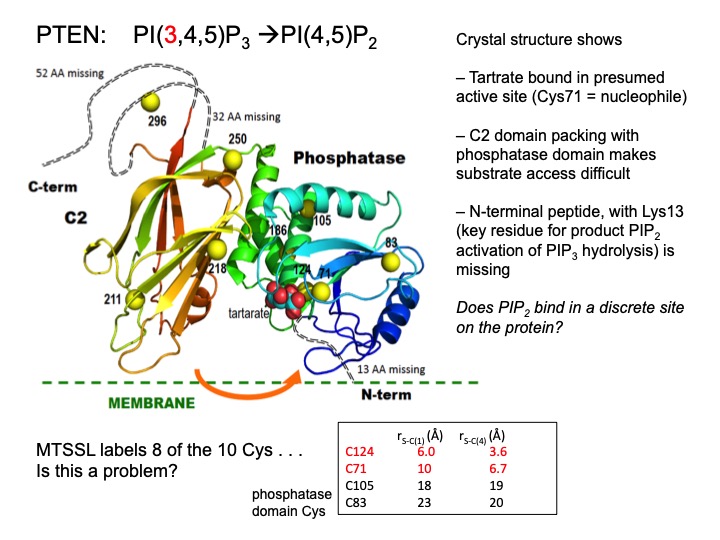
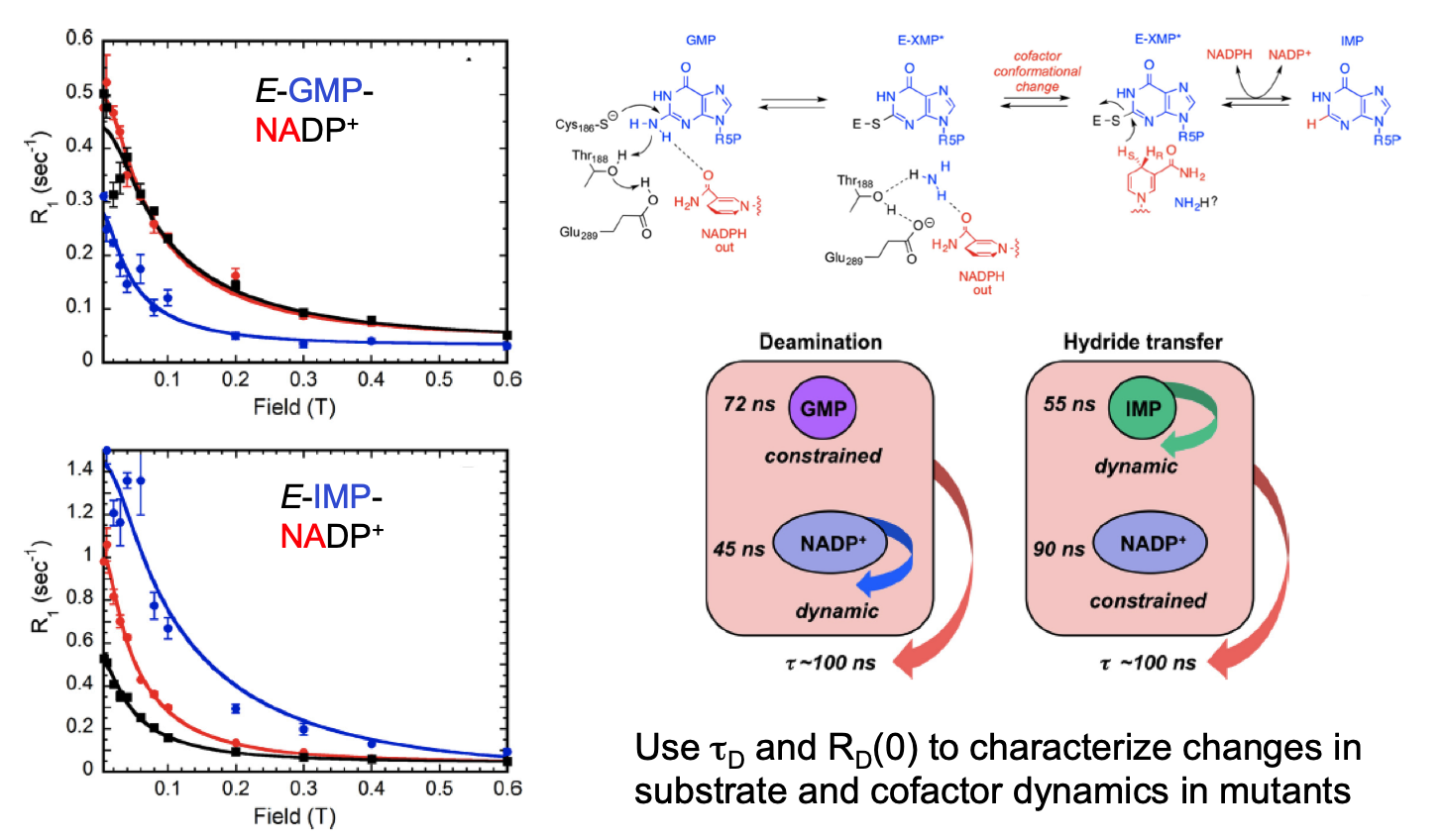
One of the best and easiest uses of Al’s Spin Spa has been to look at how moderately large macromolecules bind small molecules – for an enzyme these could be substrates, inhibitors or allosteric effectors. One can get interesting information without a spin-labeled protein. What the methodology really provides is insight into the motional behavior of the small molecule when bound. We carried out 31P field cycling with the enzyme GMPR (guanosine-5’-monophosphate reductase) – an tetramer that carries out a deamination reaction,
GMP + NADPH → IMP + NADP+
and a hydride transfer reaction. Crystal structures show that the cofactor has two distinct conformations.[Ref: Yang et al. (2015) J. Biol. Chem. 290:1592-1606] The 31P field-cycling with substrates and NADP+ (these form dead end complexes that mimic distinct catalytic steps) showed that each direction has distinct τ and R(0) values, and mutations that alter enzyme activity can be understood by determining how the NMR parameters for substrate and cofactor are affected.
The last work Al was doing was trying to understand all the features observed in the 31P and 13C field cycling profile for phospholipid vesicles. That ‘excess’ R1 bothered him. About four years ago, he thought it could be a cross-correlation between the dipolar relaxation associated with vesicle rotation and CSA. Another possibility was that it was a fast dipolar interaction where the drop at high fields was masked by the increase in the CSA R1. We had acquired 31P, 13C (carbonyls enriched), and limited 1H data for the same phospholipid in vesicles and were working on a manuscript describing arguments for each of those possibilities (for anyone who has worked on a manuscript with Al, how many versions did you go through? I referred to it as the REM (Redfield editions of manuscript) number and never got below 14 distinctly different versions the REM was usually > 25 versions).
Al was also intrigued by the CSA – he called it the transient CSA interaction - and the only phosphorus compound we examined that didn’t show measurable CSA 31P relaxation in solution was inorganic phosphate. Al was concocting a theory that water transiently protonated the phosphodiester, which would obviously change the CSA. We even managed to obtain data at much higher fields (on fixed field instruments) that showed a deviation from a square law dependence of R1 on magnetic field and could get a correlation time for the CSA relaxation interaction. That τ in the range one would expect for H2O interacting with the phosphate. Unfortunately, this wasn’t quite finished . . .
In summary, Al Redfield was a creative physicist who loved doing novel experiments – especially if it meant building something. I loved talking with him – he is sorely missed!
related works
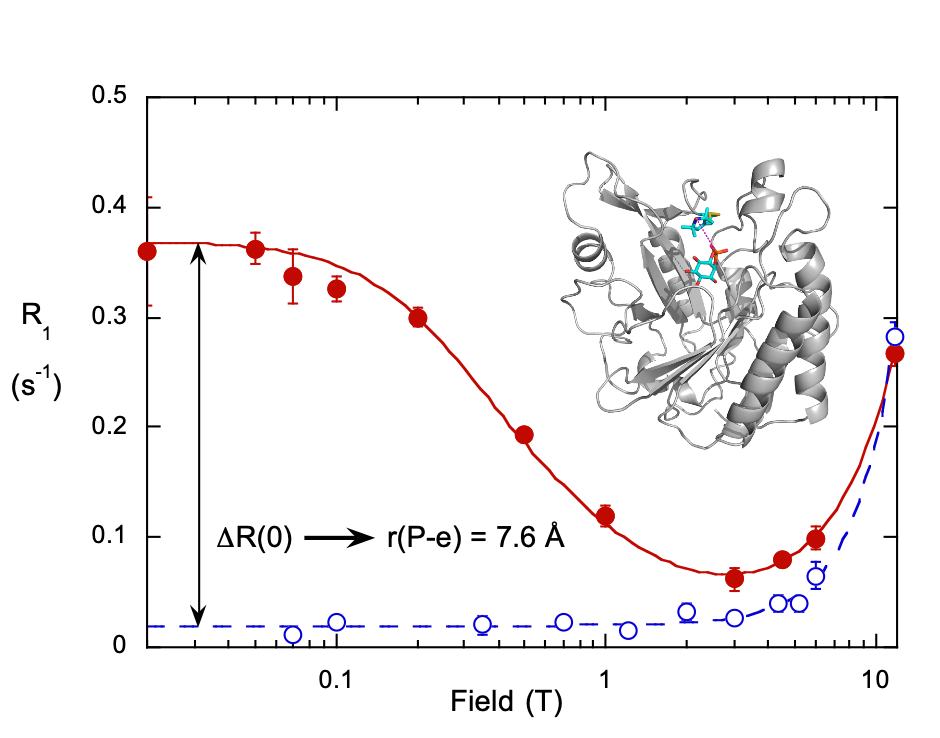
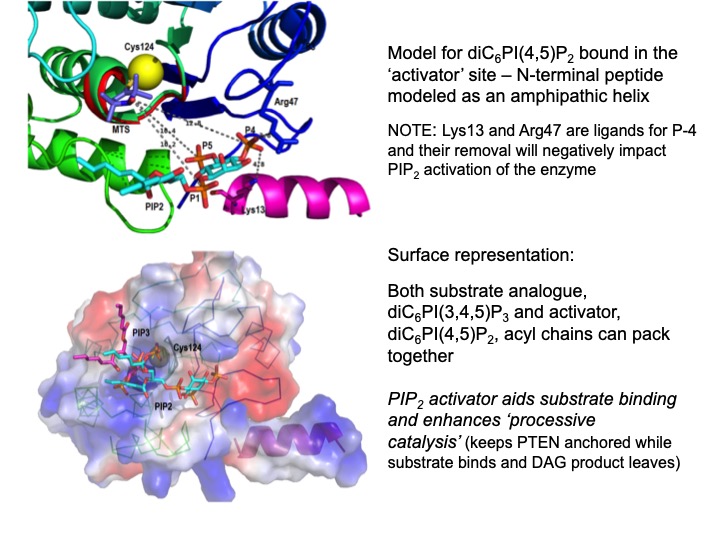
Enzymology with a Spin-Labeled Phospholipase C: Soluble Substrate Binding by 31P NMR from 0.005 to 11.7 T
Pu, Feng, Redfield, and Roberts Biochemistry 48, 8282-8284 (2009).
Monitor weak binding of ligands (e.g. cIP to PI-PLC H82C-SL) as a way of understanding interfacial enzymology (do interfaces really influence ligand affinity, Kd, or when rates increase is it because of a change in a different step . . . not binding . . . ) – use spin-labeled PLC
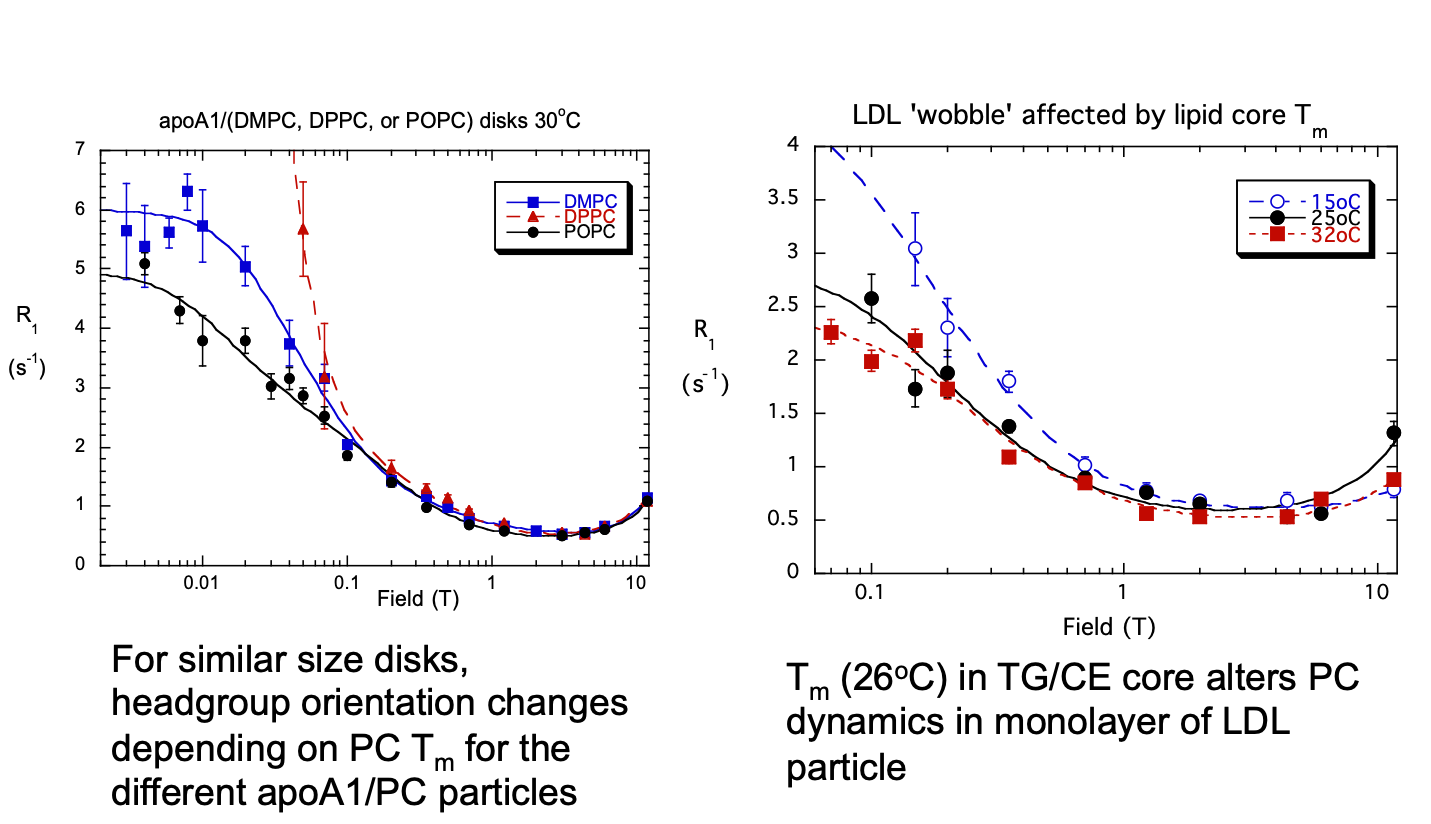
Gursky, Redfield and Roberts, unpublished results
Characterize PC dynamics and orientations in lipoproteins and model particles
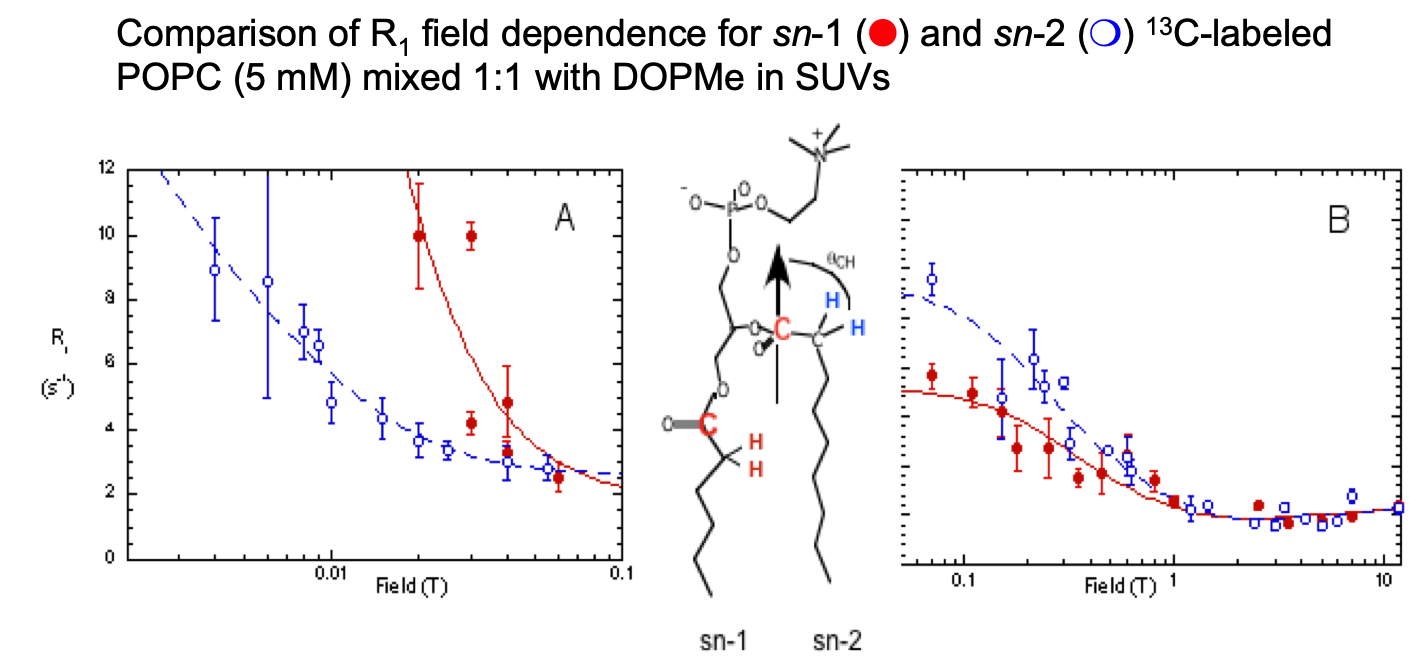
Phosphatidylcholine “Wobble” in Vesicles Assessed by High-Resolution 13C Field Cycling NMR Spectroscopy
Sivanandam, Cai, Redfield, and Roberts J. Am. Chem. Soc. 131, 3420-3421 (2009).
FC-13C-NMR of vesicles:
The two C=O have different motional characteristics (and orientations), so selectively labeled lipid can be a probe in a more complex membrane.
Substrate and Cofactor Dynamics on Guanosine Monophosphate Reductase Probed by High Resolution Field Cycling 31P NMR Relaxometry
Rosenberg, Redfield, Roberts, and Hedstrom J. Biol. Chem. 291, 22988-22998 (2016)
FC-31P-NMR for phosphorylated substrate dynamics binding to large enzymes (e.g., GMP-reductase, an octamer)