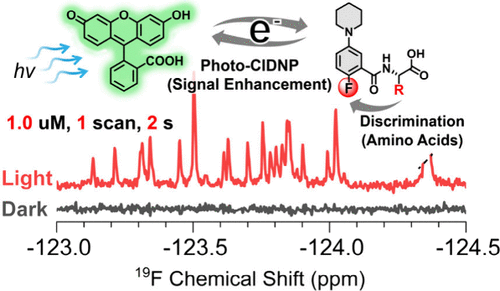
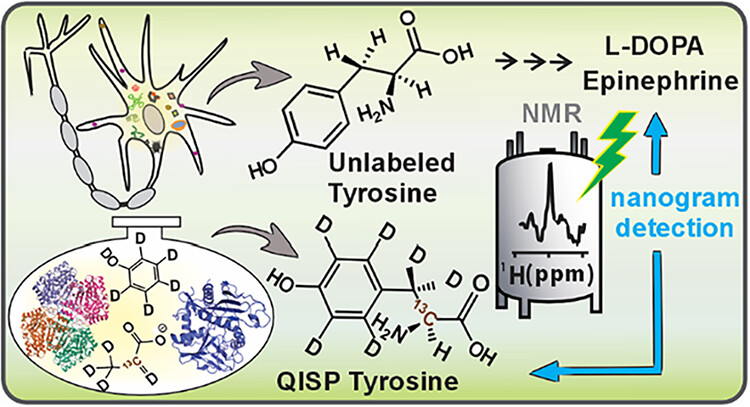
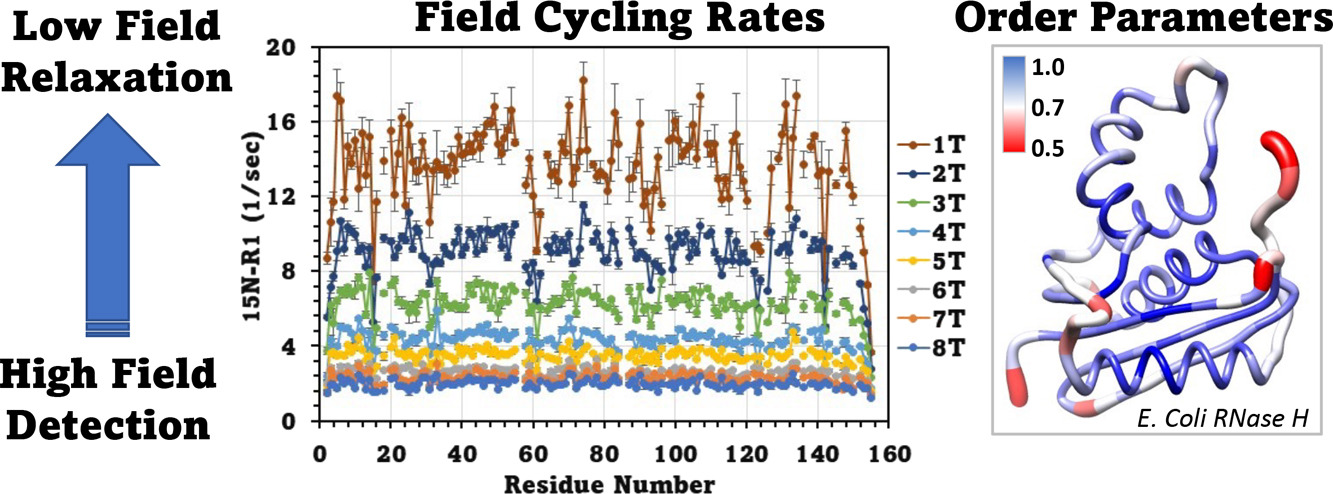
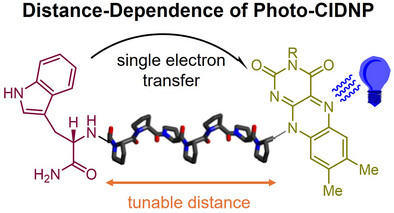
Lars T. Kuhn*, Stefan Weber, Joachim Bargon, Teodor Parella, Míriam Pérez-Trujillo*
Anal. Chem. 2024, 96, 1, 102–109
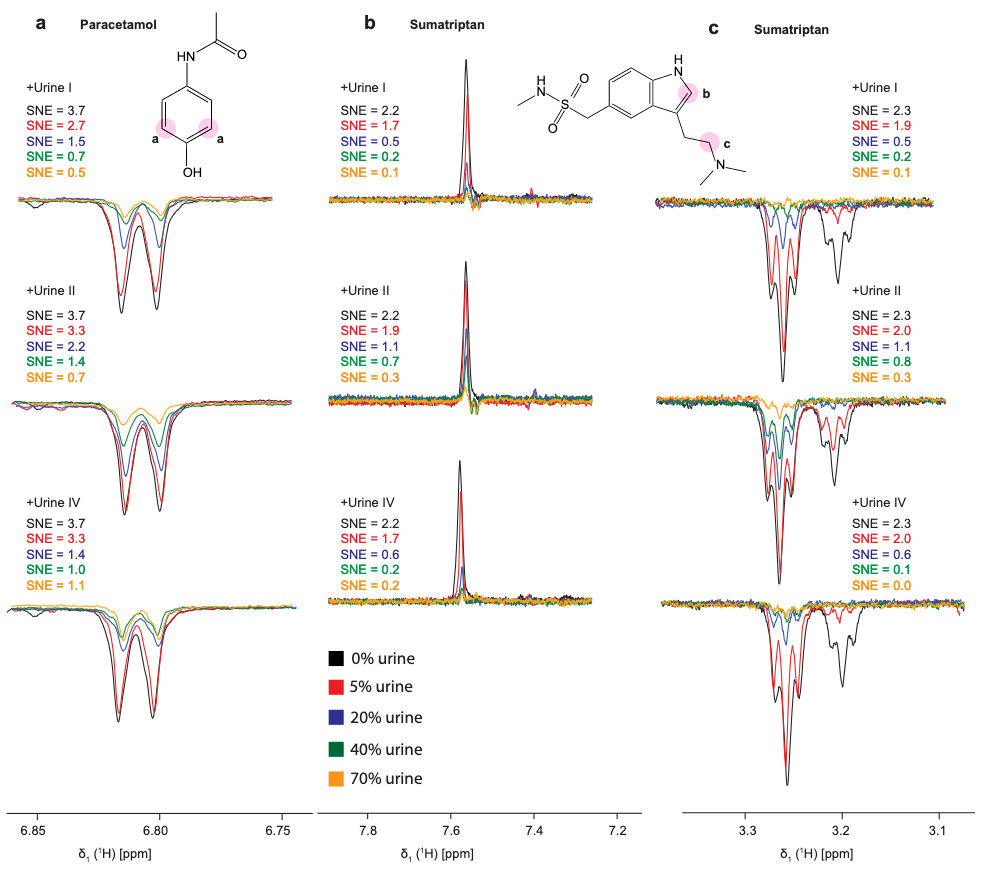
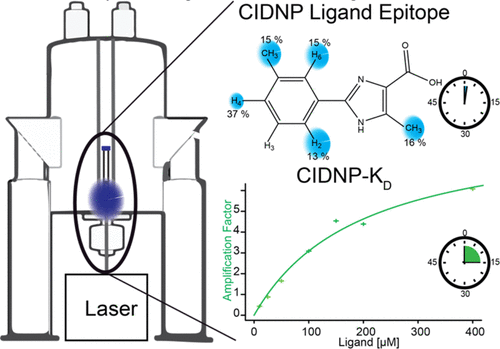
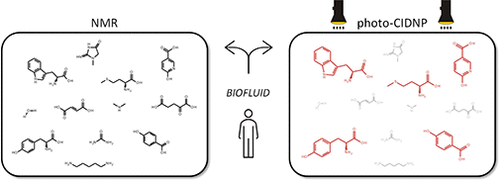
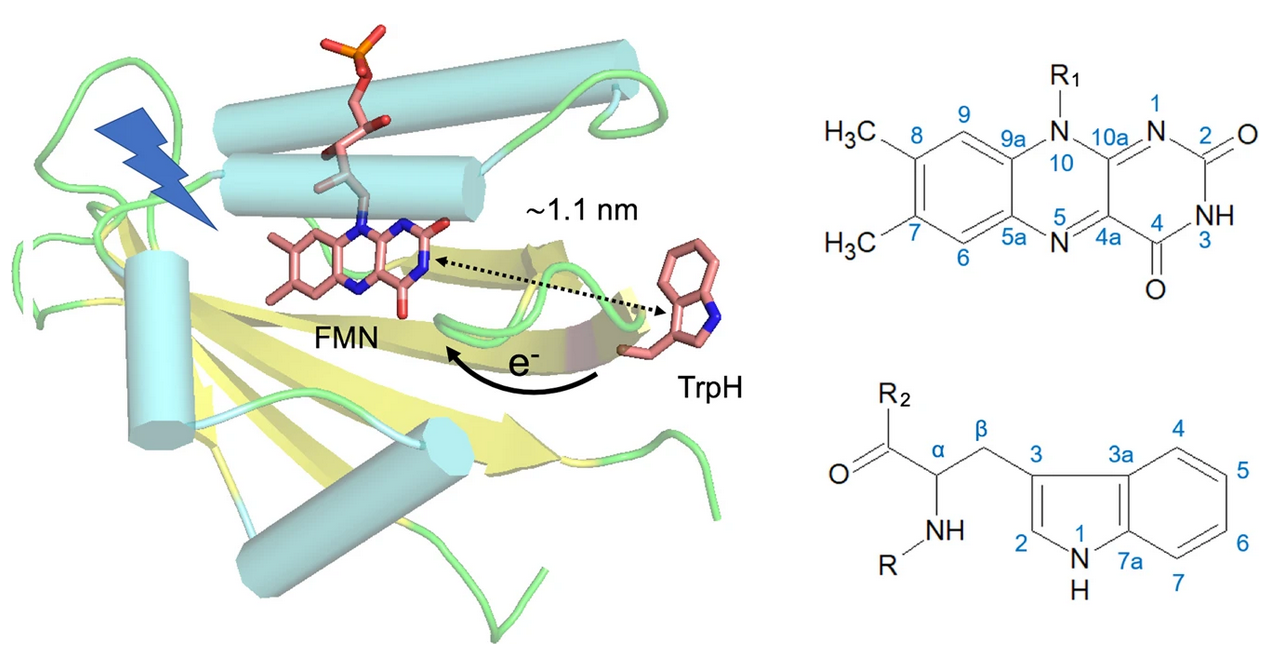
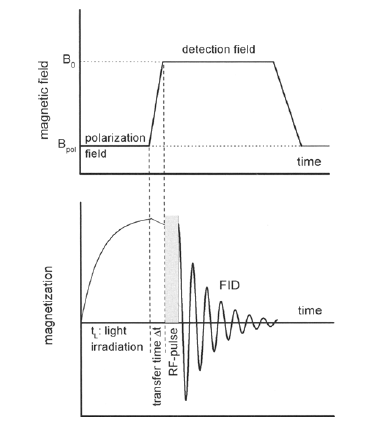
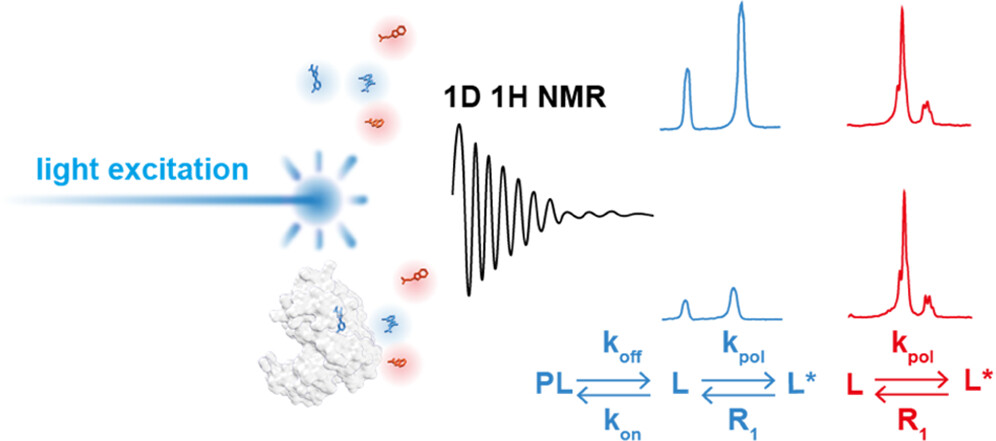
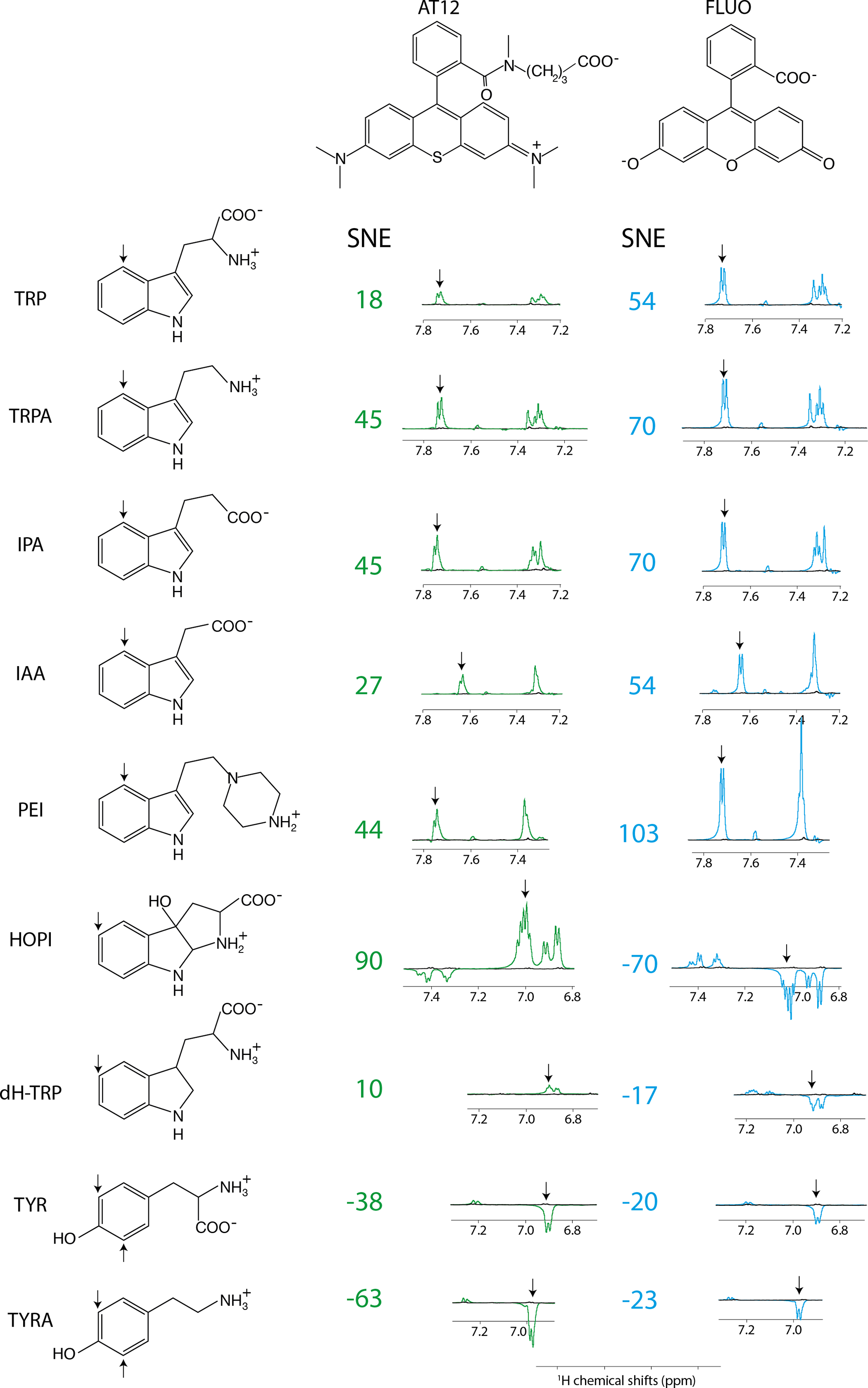
The direct and unambiguous detection and identification of individual metabolite molecules present in complex biological mixtures constitute a major challenge in (bio)analytical research. In this context, nuclear magnetic resonance (NMR) spectroscopy has proven to be particularly powerful owing to its ability to provide both qualitative and quantitative atomic-level information on multiple analytes simultaneously in a noninvasive manner. Nevertheless, NMR suffers from a low inherent sensitivity and, moreover, lacks selectivity regarding the number of individual analytes to be studied in a mixture of a myriad of structurally and chemically very different molecules, e.g., metabolites in a biofluid. Here, we describe a method that circumvents these shortcomings via performing selective, photochemically induced dynamic nuclear polarization (photo-CIDNP) enhanced NMR spectroscopy on unmodified complex biological mixtures, i.e., human urine and serum, which yields a single, background-free one-dimensional NMR spectrum. In doing this, we demonstrate that photo-CIDNP experiments on unmodified complex mixtures of biological origin are feasible, can be performed straightforwardly in the native aqueous medium at physiological metabolite concentrations, and act as a spectral filter, facilitating the analysis of NMR spectra of complex biofluids. Due to its noninvasive nature, the method is fully compatible with state-of-the-art metabolomic protocols providing direct spectroscopic information on a small, carefully selected subset of clinically relevant metabolites. We anticipate that this approach, which, in addition, can be combined with existing high-throughput/high-sensitivity NMR methodology, holds great promise for further in-depth studies and development for use in metabolomics and many other areas of analytical research.