Top > Technologies > in-situ Illumination Technologies for NMR
Overview
In situ illumination of liquid-state NMR samples opens a field for studies of light-dependent chemical or biological interactions. Among those studies, the light-driven reactions between amino acid and dye molecule, studied by photochemical induced nuclear polarization (photo-CIDNP) in solution, have been investigated to show characteristic changes in spin polarization upon various magnetic fields. The applications of photo-CIDNP, such as detecting aromatic amino acids or proteins in solution in the low micro-molar to nano-molar concentration range, are getting compelling simultaneously.
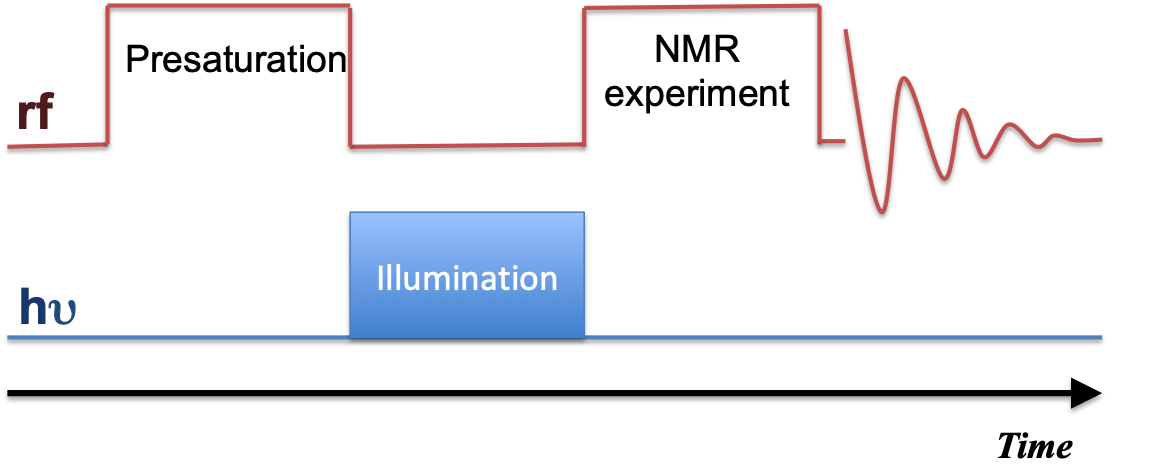
The time sequence shows how to perform the irradiation along with the NMR sequence. For low-field irradiation, the sample travels to the low field for irradiation and shuttles back immediately for pulsing and acquisition.
Due to the narrow bore of most commercial standard liquid-state NMR spectrometers, there was challenging to place a light source directly inside the magnet. Conventionally, the light source and control unit were outside of the spectrometer. The light, from lasers, or LED, was coupled through the long optical fiber to samples. The major drawback of this manner is that the long optical fiber confines the movement of the NMR sample. It would be severe for field-cycling NMR applications. For non-field-cycling studies, light homogeneity is also an issue for light coupling from the long thin optical fiber.
Hence, we have developed compact and high-precision shuttling equipment that shuttles and illuminates the sample at the stray field in a high-field NMR spectrometer to obtain high-resolution spectra containing the effect of field-dependent CIDNP, Field Cycling NMR illuminator. For non-field-cycling NMR users, we have also designed a compact illuminator installed directly on the sample tube, in-situ NMR illuminator.
Applications of Photo-CIDNP
Illuminated NMR, anchored by photo-CIDNP, offers a transformative perspective on light-matter interactions across several research domains:
- Deciphering Radical Reactions: Photo-CIDNP enhances NMR signals through polarization transfer from photoexcited radical pairs to surrounding nuclei, providing unprecedented insights into radical reaction kinetics and spatial arrangements within molecules.
- Dynamic Molecular Insights: The technique's signal enhancement capability illuminates dynamic molecular processes such as photoisomerization, capturing transient states and structural changes in photodegradation processes.
- Unveiling Hidden Pathways: Illuminated NMR uncovers "dark" reactions initiated by photochemistry. Hidden chemical pathways with transformative potential in fields like organic synthesis.
- Biomolecular Exploration: The approach advances biomolecular research by dissecting light-biomolecule interactions, vital for studying retinal proteins and their conformational changes upon light absorption.
Light Source Selection: Laser vs. LED
Irradiation Parameters and Reaction Kinetics
The efficiency of sample irradiation governs both sensitivity enhancement and the extent of photochemical reactions. The total irradiation energy Q is the product of irradiation power (P) and pulse width (W): Q = P × W. Understanding this relationship is key to controlling reaction pathways.
When Q is held constant, varying the pulse width W allows researchers to modulate reaction rates — effective when the chemical reaction timescale is comparable to or shorter than W. As pulse width approaches or exceeds the reaction timescale, longer irradiation increases the probability of energy absorption, raising the observed reaction rate. This provides a versatile handle for fine-tuning photochemical processes without changing total input energy.

The choice of light source critically impacts illuminated NMR performance. Lasers offer high irradiance and monochromaticity, but come with significant drawbacks: complex optical power control requiring photodiode feedback systems, coherence-induced interference patterns causing uneven irradiation, and limited pulse width control at high power. These constraints necessitate external placement with long optical fibers, a severe limitation for field-cycling applications.
LEDs, by contrast, provide stable and linearly controllable power output, broad wavelength coverage without coherence-induced interference, and simpler setup. Crucially, when positioned proximal to the sample, as in our compact illuminators, LEDs eliminate the fiber transmission losses that disadvantage lasers in power delivery. Modern LEDs can generate substantial power outputs rivaling those of lasers at the sample, while offering homogeneous irradiation without additional optics. For field-cycling NMR setups, where sample movement is essential, LEDs positioned close to the sample are the superior choice.
References
- Siyu Li, Shibani Bhattacharya, Ching-Yu Chou, Minglee Chu, Shu-Cheng Chou, Marco Tonelli, Michael Goger, Hanming Yang, Arthur G. Palmer, Silvia Cavagnero, Journal of Magnetic Resonance 359 (2024) 107616
- Yining Ji, Daniel A. DiRocco, Cynthia M. Hong, Michael K. Wismer, and Mikhail Reibarkh, Organic Letters 2018 20 (8), 2156-2159, DOI: 10.1021/acs.orglett.8b00391
- Dan Lehnherr, Yining Ji, Andrew J. Neel, Ryan D. Cohen, Andrew P. J. Brunskill, Junyu Yang, and Mikhail Reibarkh, Journal of the American Chemical Society 2018 140 (42), 13843-13853, DOI: 10.1021/jacs.8b08596
- Fedin, M.V., E.G. Bagryanskaya, and P.A. Purtov, Theoretical and experimental studies of chemically induced dynamic nuclear polarization kinetics in recombination of radical pairs by the method of switched external magnetic field. II. 13C CIDNP of micellized radical pairs. The Journal of Chemical Physics, 1999. 111(12): p. 5491-5502.
- Kuhn, L.T., Photo-CIDNP NMR Spectroscopy of Amino Acids and Proteins. 2013. 338: p. 229-300.
- Magin, I.M., et al., Low field photo-CIDNP in the intramolecular electron transfer of naproxen–pyrrolidine dyads. Physical Chemistry Chemical Physics, 2016. 18(2): p. 901-907.
- Seegerer, A., P. Nitschke, and R.M. Gschwind, Combined In Situ Illumination-NMR-UV/Vis Spectroscopy: A New Mechanistic Tool in Photochemistry. Angewandte Chemie International Edition, 2018. 57(25): p. 7493-7497.
- Ji, Y., et al., LED‐Illuminated NMR Spectroscopy: A Practical Tool for Mechanistic Studies of Photochemical Reactions. ChemPhotoChem, 2019. 3(10): p. 984-992.
- Grosse, S., et al., Field cycling by fast NMR probe transfer: Design and application in field-dependent CIDNP experiments. Applied Magnetic Resonance, 1999. 17(2): p. 211-225.
- Philipp Nitschke, Nanjundappa Lokesh, Ruth M. Gschwind, Combination of illumination and high resolution NMR spectroscopy: Key features and practical aspects, photochemical applications, and new concepts. Progress in Nuclear Magnetic Resonance Spectroscopy, Volumes 114–115, October–December 2019, Pages 86-134
- Lars T. Kuhn, Míriam Pérez-Trujillo, Continuous-Wave (CW) Photo-CIDNP NMR Spectroscopy: A Tutorial, Magnetic Resonance in Chemistry: 2025. 0:1–15.
- Kaptein, R. (1982). Photo-CIDNP Studies of Proteins. In: Biological Magnetic Resonance. Springer, Boston, MA.
Technical Note: Chou, C., "Illuminated NMR in Chemical Analysis", Field Cycling Communication, Issue 15 (2023).
Frequently Asked Questions
- Should I use a laser or an LED for photo-CIDNP NMR experiments?
For most photo-CIDNP applications, LEDs are the superior choice. Unlike lasers, LEDs provide stable and linearly controllable power output, homogeneous irradiation without coherence-induced interference patterns, and simpler setup without photodiode feedback systems. When positioned proximal to the sample — as in compact in-situ illuminators — LEDs eliminate fiber transmission losses and can deliver power outputs rivaling lasers at the sample. For field-cycling NMR where sample movement is essential, LEDs are the only practical option.
- What is photo-CIDNP and what can it detect?
Photo-CIDNP (photochemically induced dynamic nuclear polarization) enhances NMR signals through polarization transfer from photoexcited radical pairs to surrounding nuclei. This signal enhancement enables detection of aromatic amino acids and proteins in solution at low micromolar to nanomolar concentrations — far below conventional NMR sensitivity limits. It is also used to study radical reaction mechanisms, photoisomerization, and light-biomolecule interactions.
- How does irradiation pulse width affect photo-CIDNP experiments?
The total irradiation energy Q = P × W (power × pulse width) governs photochemical reaction kinetics. When total energy Q is held constant, varying the pulse width W modulates reaction rates — particularly effective when the chemical reaction timescale is comparable to or shorter than W. This provides a handle for fine-tuning photochemical processes without changing total input energy, allowing researchers to control reaction pathways independently of power levels.
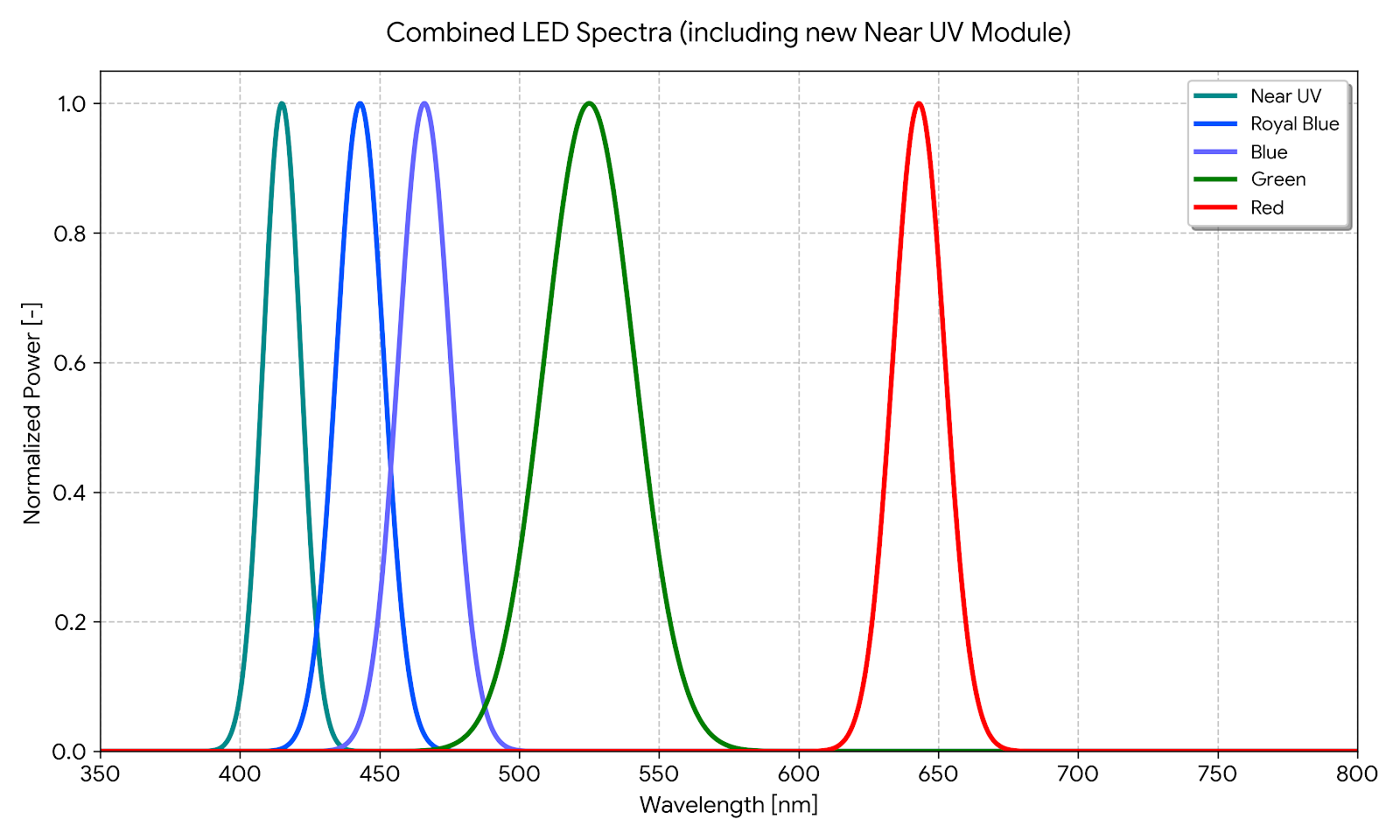
- What LED wavelengths are recommended for in-situ NMR photocyclization experiments?
Wavelength selection depends on the absorption spectrum of the photosensitizer or substrate. For flavin-based photo-CIDNP, 445–450 nm blue LEDs are standard. For [2+2] photocycloadditions commonly monitored by in-situ LED-NMR (as in Lehnherr et al., JACS 2018), 455 nm blue LEDs are typically used. For reactions requiring near-UV activation, 365–405 nm LEDs are appropriate. The FCNMR illuminator supports interchangeable LED modules across the UV-visible range to accommodate different photochemical systems. For a comprehensive reference of photosensitizer–analyte pairs and their excitation wavelengths, see our Database of Dyes for Photo-CIDNP.
